I’m sure you’ve seen more than one article pronouncing that meat causes cancer. Depending on the source, the article might have a plant-based diet lean and demonize all meat, or merely throw red meat or deli meats under the cancer bus. I know I get asked nearly daily to rebut some article or other on the subject.
But, the truth is that there really are a lot of studies out there linking meat consumption (especially red meat) to cancer—in human populations, in animal models, and even in some controlled trials focusing on pre-cancerous changes in the human body. Hardly a month goes by without seeing at least one of these studies pop up in the headlines and make the rounds on social media. These studies seem to buoy critics of the Paleo diet and low-carb diets alike, while providing fuel for harassment from family members who just don’t get how we eat (or why).
Within the Paleo community, there is a strong tendency to dismiss these scientific studies as being irrelevant or poorly conducted. The most standard response is “But the study didn’t use grass-fed and organic meat!” and then we go on our merry way with the assumption that the results don’t apply to us (and we tell our family members so!). Then, there’s always the more general anti-science sentiments, dismissing the relevance of a study based on perceived design flaws or legitimate study limitations. As you can probably imagine, dismissing science because it doesn’t conform to our established beliefs is not something I endorse.
Studies linking meat consumption to cancer are relevant to the Paleo community and are worthy of discussion. So, let’s do that.
Researchers have uncovered several mechanisms linking cancer with components of meat that have nothing to do with an animal’s diet or antibiotic exposure, including heme iron, specific proteins, other specific molecules, and heat-induced mutagens. These are things that exist in meat whether it’s conventional or grass-fed or wild game. That means that organic grass-fed meat, while it promotes health in other ways (better fats, more micronutrients), still has the capacity to increase cancer risk. So, instead of dismissing the meat and cancer research as irrelevant, let’s take the time to objectively look at how it affects us.
But, don’t get scared over to vegetarianism just yet! When we take a closer look at these studies, we see something extraordinarily interesting: the link between meat and cancer tends to disappear once the studies adjust for vegetable intake. Even more exciting, when we examine the mechanistic links between meat and cancer, it turns out that many of the harmful (yes, legitimately harmful!) components of meat are counteracted by protective compounds in plant foods.
Said more simply: YES, meat does cause cancer! …IF you aren’t eating your veggies!
This is yet another reason why the Paleo diet can be considered a plant-based diet thanks to its focus on eating a large amount of vegetables! See The Diet We’re Meant to Eat, Part 1: Evolution & Hunter-Gatherers, The Diet We’re Meant to Eat, Part 2: Physiological & Biological Evidence, The Diet We’re Meant to Eat, Part 3: How Much Meat versus Veggies? and The Importance of Vegetables.
Let’s dive into the mechanistic details!
Heme
A major mechanism linking meat to cancer involves heme, the iron-containing compound that gives red meat its color (in contrast to the nonheme iron found in plant foods). Where heme becomes a problem is in your gut: the cells lining your digestive tract metabolize it into cytotoxic compounds (meaning toxic to living cells), which can then damage your colonic mucosa, cause cell proliferation, and increase fecal water toxicity—all of which raise cancer risk. Yikes! In fact, part of the reason red meat shows up linked with cancer far more often than white meat could be due to their differences in heme content: white meat (poultry and fish) contains much, much less of it.
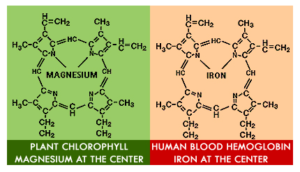 Here’s where vegetables come to the rescue! Chlorophyll, the pigment in plants that makes them green, has a molecular structure that’s very similar to heme. As a result, chlorophyll can block the metabolism of heme in your intestinal tract and prevent those toxic metabolites from forming. Instead of turning into harmful byproducts, heme ends up metabolized into inert compounds that are no longer toxic or damaging to your colon. Animal studies have demonstrated this effect in action: one study on rats showed that supplementing a heme-rich diet with chlorophyll (in the form of spinach) completely suppressed the pro-cancer effects of heme. All the more reason to eat a salad with your steak!
Here’s where vegetables come to the rescue! Chlorophyll, the pigment in plants that makes them green, has a molecular structure that’s very similar to heme. As a result, chlorophyll can block the metabolism of heme in your intestinal tract and prevent those toxic metabolites from forming. Instead of turning into harmful byproducts, heme ends up metabolized into inert compounds that are no longer toxic or damaging to your colon. Animal studies have demonstrated this effect in action: one study on rats showed that supplementing a heme-rich diet with chlorophyll (in the form of spinach) completely suppressed the pro-cancer effects of heme. All the more reason to eat a salad with your steak!
Nutrivore Weekly Serving Matrix
An easy-to-use and flexible weekly checklist
to help you maximize nutrient-density.
The Weekly Serving Matrix is very helpful! I’ve been eating along these lines but this really helps me know where to focus vs. which foods serve a more secondary role. It’s super helpful and has taken a lot of worry out of my meal planning. Thanks!
Jan
L-Carnitine and TMAO
L-carnitine is an amino acid that’s particularly abundant in red meat (another candidate for why red meat seems to disproportionately increase risk of cancer compared to other meats). When you consume L-carnitine, your intestinal bacteria metabolize it into a compound called trimethylamine (TMA). From there, the TMA enters your bloodstream and gets oxidized by your liver into yet another compound, trimethylamine-N-oxide (TMAO). This is the one we should pay attention to!
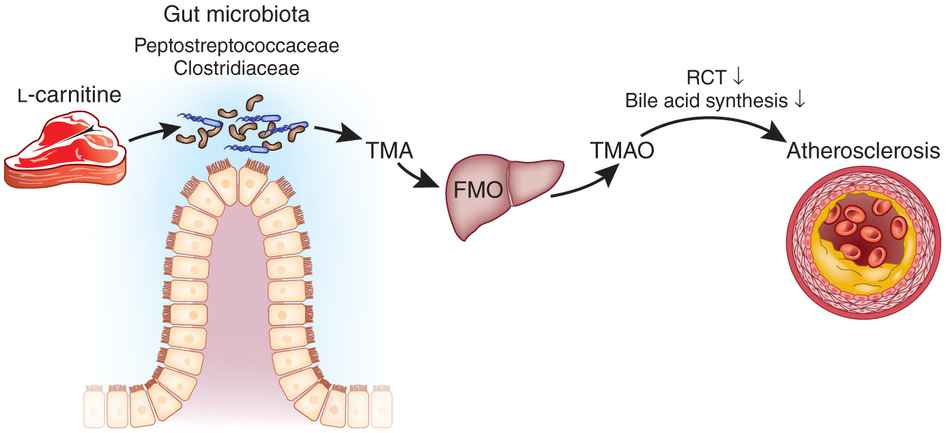 TMAO has been strongly linked to cancer and heart disease, possibly due to promoting inflammation and altering cholesterol transport. Having high levels of it in your bloodstream could feasibly be a major risk factor for some chronic diseases. So, is this the nail in the coffin for meat eaters?
TMAO has been strongly linked to cancer and heart disease, possibly due to promoting inflammation and altering cholesterol transport. Having high levels of it in your bloodstream could feasibly be a major risk factor for some chronic diseases. So, is this the nail in the coffin for meat eaters?
Not so fast! An important study on this topic was published in 2013 in Nature Medicine, and sheds light on what’s really going on. This paper had quite a few components (discussed in detail in a fascinating blog post by Jeff Leach of the Human Food Project), but one of the most interesting has to do with gut bacteria. Basically, it turns out that the bacteria group Prevotella is a key mediator between L-carnitine consumption and having high TMAO levels in your blood. In this study, the researchers found that participants with gut microbiomes dominated by Prevotella were the ones who produced the most TMA (and therefore TMAO, after it reached the liver) from the L-carnitine they ate. Those with microbiomes high in Bacteroides rather than Prevotella saw dramatically less conversion into TMA and TMAO.
Guess what Prevotella loves to snack on? Grains! It just so happens that people with high Prevotella levels tend to be those eating grain-based diets (especially whole grain), since this bacterial group specializes in fermenting the type of polysaccharides abundant in grain products. (For instance, we see extremely high levels of Prevotella in populations in rural Africa that rely on cereals like millet and sorghum.) At the same time, Prevotella doesn’t seem to be associated with a high intake of non-grain plant sources, such as fruit and vegetables.
So, is it really the red meat that’s a problem… or is it the meat in the context of a grain-rich diet? Based on the evidence we have so far, it seems that grains (and the bacteria that love to eat them) are a mandatory part of the L-carnitine-to-TMAO pathway. Ditch the grains, and your gut will become a more hospitable place for red meat!
Neu5GC
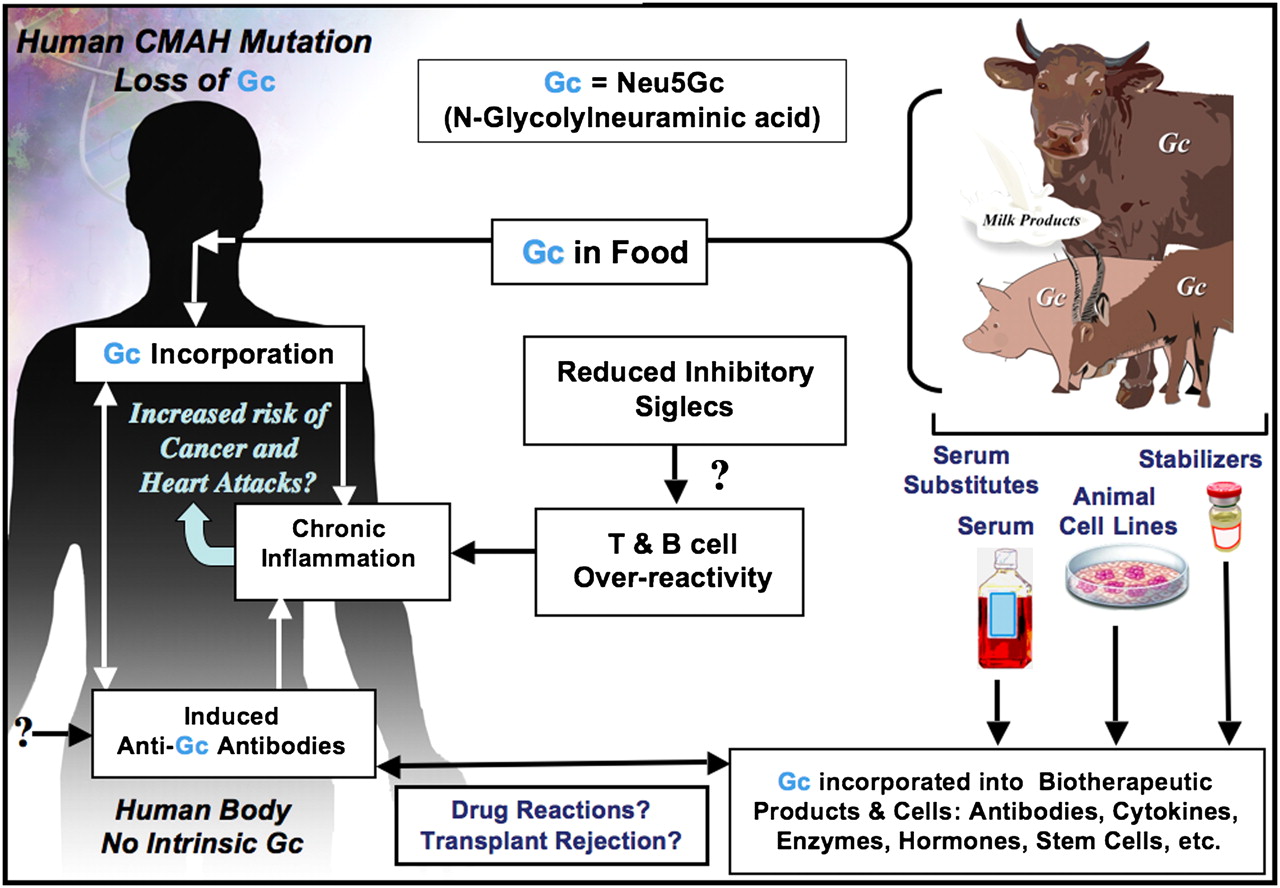
Another lesser-known mechanism behind the meat and cancer link involves a sialic acid molecule called Neu5GC, which is found abundantly in red meat. The human body can’t produce Neu5GC (instead, we make a different sialic acid molecule called Neu5AC), but we can incorporate Neu5GC into our cell membranes if we ingest it from food. There’s just one problem: since Neu5GC is a foreign molecule, our bodies can launch an immune response against the Neu5GC we’ve incorporated, leading to the production of antibodies and inflammation. That process appears to be involved in raising cancer risk. (In fact, mice studies designed to simulate what happens in the human body are showing that after long-term exposure, Neu5GC accumulates in tumors and significantly raises the number of carcinomas that form.)
The question then becomes why do we form antibodies against Neu5GC at all? Not everyone does, and it may be something that happens in an over-stimulated or dysfunctional immune system, similar to the way we form antibodies against our own tissues in autoimmune disease. In this case, nutrient sufficiency, gut health, adequate sleep, stress management and activity are all important inputs to prioritize (all discussed in detail in The Paleo Approach). And it is thus no surprise that nutrient deficiency, gut dysbiosis, poor sleep, high stress and inactivity have all been linked to increased cancer risk.
Do vegetables play a role here too? Unfortunately, that’s something researchers are still exploring—and we can’t say yet how vegetable intake influences the potential Neu5GC-cancer connection. But, we do know from a giant range of studies that vegetables exert cancer-protective properties through a number of pathways, and every study that corrects for vegetable intake finds the meat and cancer link to disappear. High vegetable intake benefits the immune system in many ways too. So, we can make an educated guess at this point and say that whatever risk Neu5GC may create, vegetables appear to negate it.
Mutagens from Cooking
Okay, a juicy steak on the grill tastes amazing. I think we can all agree on that. But, what’s not amazing is the molecular effect high-temperature cooking has on meat. Harsh cooking methods like grilling and frying can generate compounds called heterocyclic amines, or HAs (formed from reactions between amino acids and creatine in muscle meat) and polycyclic aromatic hydrocarbons, or PAHs (formed when meat drippings hit open fire and cause PAH-containing flames to rise up, in turn coating the meat with PAHs). Both of these compounds are known to be mutagenic, causing changes in DNA that may increase your risk of cancer. Plenty of animal experiments and human population studies point towards HAs and PAHs being major players in the cancer correlations we see with meat.
Although we’re not 100% sure how dramatically HAs and PAHs raise cancer risk in humans (it’s really hard to measure people’s exact intake and correlate it with disease incidence over time), it’s safe to say these compounds aren’t harmless, and we should try to minimize our exposure to them. One way to do that, of course, is to stick with gentle cooking methods (hello, crockpot!) instead of charring our meat to smithereens. But, guess what else can counteract meat mutagens? It probably won’t come as a surprise that the answer, again, is vegetables!
Multiple studies have shown that indoles, a class of phytochemicals abundant in cooked or crushed/chewed crucifers (like broccoli, cauliflower, Brussels sprouts, and cabbage), can suppress the tumors induced by PAHs and HAs, as well as totally change the way your body metabolizes these mutagens to make them less harmful. One study found that feeding people a diet containing 500 grams of crucifers per day (in the form of broccoli and Brussels sprouts) reduced the formation of toxic metabolites from a particularly dangerous HA called 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (try saying THAT five times fast!). Basically, it looks like we can protect ourselves from some of the damage these mutagens cause by making sure we eat plenty of cruciferous vegetables. Let’s fire up the barbecue! (and make a shaved Brussels salad!)
Nitrites and Nitrosamines
When we consume a meat cured with nitrites (including those naturally-occurring in celery extract or powder), nitrosamines can be formed via a chemical reaction with a variety of proteins in our bodies. About 90% of the 300 or so studied nitrosamines are carcinogenic and it has been postulated that this is the mechanism behind the (albeit weak) statistical link between cured meat consumption and incidence of some types of cancer (most notably gastric cancer, thyroid cancer and glioma). Two meta-analyses confirm that excessive consumption of nitrites or a specific nitrosamine called N-nitrosodimethylamine (NDMA) increases risk of stomach cancer (counterintuitively, increased consumption of nitrates, which can also form nitrosamines, decreases risk of stomach cancer). Another meta-analysis confirmed a small increase in risk of thyroid cancer can glioma with high nitrite intake, but no increase in risk of bladder cancer, breast cancer, colorectal cancer, esophageal cancer, non-Hodgkin lymphoma, ovarian cancer, pancreatic cancer, prostate cancer, and renal cell cancer.
Despite the relatively weak connection between nitrite consumption and cancer risk, this is yet another mechanism linking meat and cancer where vegetables can come to the rescue! Vitamin C and many phenolic compounds (see Polyphenols: Magic Bullet or Health Hype?) inhibits the chemical reactions that produce nitrosamines, explaining why any link between cured meat and cancer disappears in people who consume even a modest amount of fresh vegetables and fruit.
But What About the Inuit?
A common argument for the claim that red meat can cause cancer when you don’t eat enough plant foods is, “What about the Inuit?” After all, didn’t they stay cancer-free on a diet that was nothing but meat?
Actually, this is a myth! The Inuit (on their traditional diet) definitely had a huge intake of animal foods, but they also went to great lengths to gather amazingly nutrient-dense plants whenever they were available (as well as preserve them for year-round use). Those included kelp, algae, and other seaweeds (which are super high in chlorophyll), plankton from whales’ stomachs, fireweed, sorrel grass, flower blossoms preserved in seal oil, mosses, a variety of berries, ground nuts, lichens, willow leaves, sourdock, scurvygrass, roseroot, numerous other weeds and greens, tubers, starchy corms (which the Inuit collected from caches made by tundra mice and voles), and the partially digested stomach contents of caribou and other land animals (considered a delicacy, and usually consisting of a variety of lichens, blueberry leaves and shoots, horsetails, grasses, birch, willow, and other plants the animals had grazed on). Whew! That’s quite a list. And, many of those wild foods have a much higher micronutrient content than the cultivated vegetables we get from the store, so a little went a long way.
 Plus, the Inuit traditionally fermented large quantities of their greens, berries, and roots as a way of preserving them. If you’ve read my posts on fermentation (check out my latest one here!), you know that fermented foods can have major anti-cancer properties, and might offset the increased risk from red meat by shifting the gut microbiome and protecting the colon from dangerous precancerous mutations.
Plus, the Inuit traditionally fermented large quantities of their greens, berries, and roots as a way of preserving them. If you’ve read my posts on fermentation (check out my latest one here!), you know that fermented foods can have major anti-cancer properties, and might offset the increased risk from red meat by shifting the gut microbiome and protecting the colon from dangerous precancerous mutations.
What’s more, we can’t rule out the possibility that the Inuit evolved a unique gut ecology that helps them cope with their diet. We already know they evolved a genetic CPT1A mutation that prevents most Inuit from entering ketosis, something not found in those of us descended from non-arctic populations. Could something similar have happened with the Inuit gut microbiome? It’s definitely possible.
Bottom line: despite a high meat intake, the Inuit were also eating a huge variety of protective plant foods (and receiving the perks of fermentation), so they really don’t invalidate the red meat and cancer connection.
Does This Mean It’s Better to Be Vegetarian?
Not at all! The point of this post isn’t to scare you away from meat, but to emphasize that both plant foods and animal foods play an important (and symbiotic) role in the human diet. Without the protective effects of plant foods, some components of animal products—meat, in this case—can become legitimately harmful and raise your risk of diseases like cancer. But, animal products play an equally valuable role for our health, providing nutrients that are low or absent from plant foods (like B12, preformed vitamin A, zinc, highly absorbable iron, easiest-to-digest protein, and complete protein).
The danger comes when we have an imbalance between our intake of plant foods and animal products—tilting too far in the direction of either carnivore or vegan. The key is to enjoy nutrient-dense items from both the plant and animal kingdoms that lead to healthy gut flora, an excellent micronutrient status, and happy taste buds.
While a meat-heavy diet may increase cancer risk, eating lots of veggies mitigates it. When you eat both meat and a variety of vegetables (at least 5 veggie servings a day), the rationale for concerns falls short.
Yes, I just concluded what other Paleo peeps have been saying all along: meat is an essential component of a healthy diet for optimal health. But, I think it’s important to emphasize that a detailed reading of the science emphasizes just how important it is for high vegetable consumption to be another essential component of said healthy diet for optimal health.
So go ahead and eat that steak, just steam some broccoli and make a salad to go with it.
Citations
Balder HF, et al. “Heme and chlorophyll intake and risk of colorectal cancer in the Netherlands cohort study.” Cancer Epidemiol Biomarkers Prev. 2006 Apr;15(4):717-25.
Bennett J & Rowley S. “Chapter 5: Gathering.” Uqalurait: An Oral History of Nunavut. 2004: McGill-Queen’s University Press. pp. 84–85.
Clement FJ, et al. “A Selective Sweep on a Deleterious Mutation in CPT1A in Arctic Populations.” Am J Hum Genet. 2014 Oct 23;95(5):584-589.
Combet E et al. Dietary phenolic acids and ascorbic acid: Influence on acid-catalyzed nitrosative chemistry in the presence and absence of lipids. Free Radic Biol Med. 2010 Mar 15;48(6):763-71.
De Filippo C, et al. “Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa.” Proc Natl Acad Sci U S A. 2010 Aug 17;107(33):14691-6.
Kang DW, et al. “Reduced incidence of Prevotella and other fermenters in intestinal microflora of autistic children.” PLoS One. 2013 Jul 3;8(7):e68322.
Kuhnlein H & Turner N. “Traditional Plant Foods of Canadian Indigenous Peoples: Nutrition, Botany and Use.” (Food and Nutrition in History and Anthropology). 2001: Taylor and Francis.
Murray S, et al. “Effect of cruciferous vegetable consumption on heterocyclic aromatic amine metabolism in man.” Carcinogenesis (2001) 22 (9): 1413-1420.
Price W. “Chapter 5: Isolated and Modernized Eskimos.” Nutrition and Physical Degeneration. 1939: Paul B. Hoeber, Inc.
Koeth RA, et al. “Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis.” Nat Med. 2013 May;19(5):576-85.
Samraj AN, et al. “A red meat-derived glycan promotes inflammation and cancer progression.” PNAS January 13, 2015 vol. 112 no. 2 542-547.
Song P et al. Dietary Nitrates, Nitrites, and Nitrosamines Intake and the Risk of Gastric Cancer: A Meta-Analysis. Nutrients. 2015 Dec 1;7(12):9872-95.
Tangvoranuntakul P, et al. “Human uptake and incorporation of an immunogenic nonhuman dietary sialic acid.” Proc Natl Acad Sci U S A. 2003 Oct 14; 100(21): 12045–12050.
de Vogel J, et al. “Green vegetables, red meat and colon cancer: chlorophyll prevents the cytotoxic and hyperproliferative effects of haem in rat colon.” Carcinogenesis 2005;26:387–93.
Walters D, et al. “Cruciferous vegetable consumption alters the metabolism of the dietary carcinogen 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) in humans.” Carcinogenesis 2004;25(9):1659-1669.
Wattenberg L & Loub W. “Inhibition of Polycyclic Aromatic Hydrocarbon-induced Neoplasia by Naturally Occurring Indoles.” Cancer Res (1978) 38: 1410.
Wu GD, et al. “Linking long-term dietary patterns with gut microbial enterotypes.” Science. 2011 Oct 7;334(6052):105-8.
Xie L, et al. Association between dietary nitrate and nitrite intake and sitespecific cancer risk: evidence from observational studies. Oncotarget. 2016 Aug 30;7(35):56915-56932.
Zhang FX, et al. Association Between Nitrite and Nitrate Intake and Risk of Gastric Cancer: A Systematic Review and Meta-Analysis. Med Sci Monit. 2019 Mar 9;25:1788-1799







 TPV Podcast, Episode 157, Skin Health
TPV Podcast, Episode 157, Skin Health