I was asked by healthline.com to comment on a new study from researchers at Lund University in Sweden that was published earlier this month in the journal PLoS ONE. The study is entitled “Intestinal Barrier Dysfunction Develops at the Onset of Experimental Autoimmune Encephalomyelitis, and Can Be Induced by Adoptive Transfer of Auto-Reactive T Cells“. Yep, that’s a mouthful, but hang in there. This study is absolutely fascinating and is very relevant to everyone battling autoimmune disease. It’s so interesting that I’m devoting an entire blog post to it!
The study was performed in a well-established mouse model of multiple sclerosis. Now, before you get all dismissive because it wasn’t performed in humans, let me assure you that animal models of human disease are extremely powerful tools for understanding the molecular details of disease. This particular model of multiple sclerosis is called experimental autoimmune encephalomyelitis, or EAE, and it is the same model that was used in the vast majority of basic science research of multiple sclerosis performed to date. In fact, most of what we know about multiple sclerosis is thanks to this experimental model.
The purpose of the study was to investigate whether or not increased intestinal permeability, or what we more colloquially refer to as a “leaky gut”, is present in EAE (the experimental model of multiple sclerosis) and, if it is, to understand when it develops and what’s causing it.
But, first, let’s take a step backward.
What exactly is a “leaky gut”?
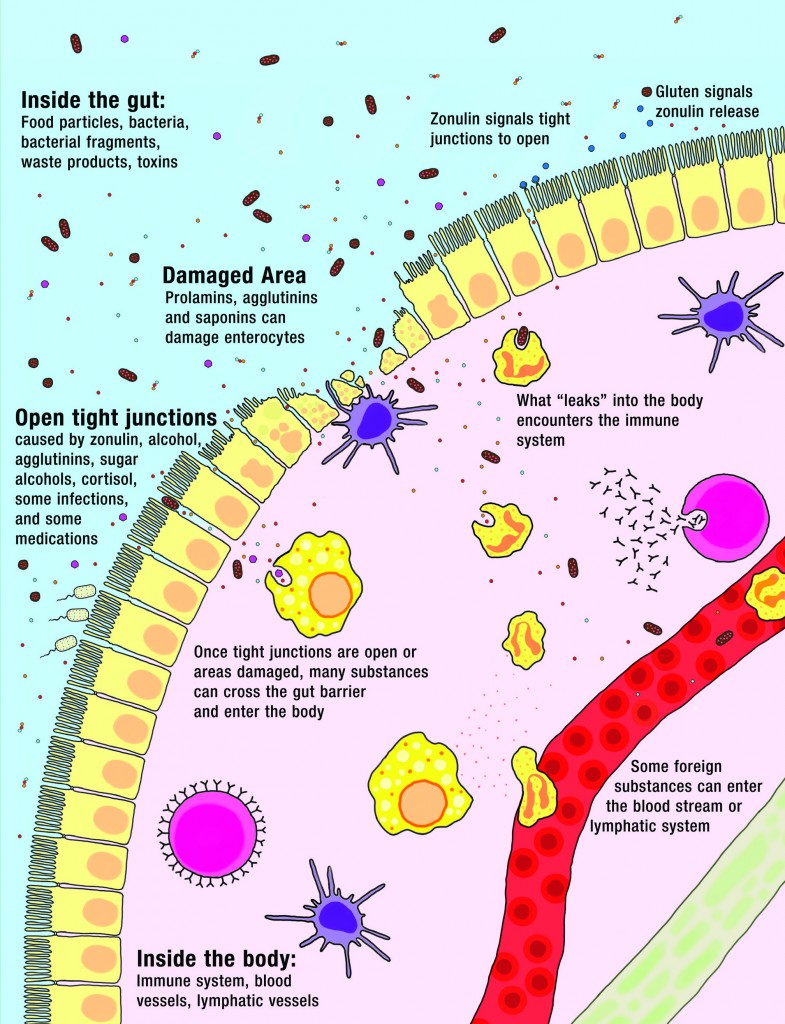
The gut is a barrier between the inside of your body and the outside world. Yes, as unintuitive as it may be, the stuff inside your digestive tract is actually outside your body. But, the gut is a very unique barrier. Its job is to let important nutrients inside the body while keeping everything else out. This makes it a highly selective semi-permeable barrier. Nutrients enter the body through a variety of tightly controlled mechanisms.
What forms this highly selective semi-permeable barrier is a single layer of highly specialized cells called enterocytes. And right on the other side of that barrier is 80% of our body’s immune systems, acting as a sentinel, ready to attack anything that might try to cross the barrier.
A leaky gut, or more technically “increased intestinal permeability”, means things can get across the gut barrier that aren’t supposed to. This happens when either the enterocytes are damaged or the complex structures that glue the enterocytes to each other are damaged. What leaks into the body isn’t big chunks of food, but a variety of small substances—like incompletely digested proteins, bacteria or bacterial fragments, infectious organisms, and waste products—which all stimulate the immune system on the other side. Some substances cause generalized bodywide inflammation (for example, bacterial fragments from those good bacteria that live in our digestive tracts but are supposed to stay there can stimulate inflammation which can then travel throughout the body). Some stimulate targeted attacks by the immune system (for example, a food intolerance or allergy could result from incompletely digested food proteins leaking into the body). The many symptoms and health conditions related to leaky gut are caused by this stimulation of the immune system.
The link between leaky gut and autoimmune disease
A leaky gut is present in every autoimmune disease in which it has been tested, including in people with rheumatoid arthritis, ankylosing spondylitis, inflammatory bowel disease (Crohn’s and ulcerative colitis), celiac disease, multiple sclerosis, and type 1 diabetes.
It’s intuitive that autoimmune diseases that attack the tissues of the gut, such as inflammatory bowel disease and celiac disease, would go hand-in-hand with a leaky gut. What’s important is that autoimmune diseases that attack tissues no where near the gut barrier, like the myelin sheaths (a protective covering that surrounds nerves of the central nervous system, i.e., the brain and spinal cord) being attacked in multiple sclerosis or the joint tissue being attacked in rheumatoid arthritis, also go hand-in-hand with a leaky gut. This implies a more fundamental role for a leaky gut in autoimmune disease.
In fact, many studies have shown that increased intestinal permeability (leaky gut)precedes the development of autoimmune disease (and in some cases can predict an oncoming flare). Yes, all the current science, including this new paper, show that a leaky gut comes first.
This is a quote from this new paper:
Recent observations in humans and in a variety of animal models indicate that an increased intestinal permeability (IP), often referred to as a “leaky gut”, is playing a pathogenic role not only in development of gastrointestinal disorders like inflammatory bowel disease (IBD) and celiac disease, but also in systemic autoimmune diseases, like type 1 diabetes (T1D).
This collection of evidence has lead many researchers and medical professionals (including me) to postulate that a leaky gut is a prerequisite for autoimmune disease to develop. This means that developing a leaky gut is part of the pathogenesis of autoimmune disease. How would this work? Because 80% of the immune system is housed in the tissues in and surrounding the gut, the development of a leaky gut stimulates to the immune system to go into overdrive. When you combine this stimulation with nutrient deficiencies (so the nutrients required for immune regulation aren’t sufficiently available), genetic predisposition (which mean the immune system is more easily stimulated or less easily regulated), and the unfortunate yet common accident of autoantibody formation (antibodies that accidentally target proteins in the human body instead of invading microorganisms), you get autoimmune disease.
What causes this leaky gut to develop? Well, until now, I and many others have postulated that this leaky gut can come from a variety of causes, from infection, to gut dysbiosis, to diet factors (like alcohol, the presence of grains in the diet, food allergies and sensitivities), to medications (like NSAIDs), to lifestyle factors (like chronic stress, consistently getting inadequate sleep, and over-activity). The mechanisms of how each of these can cause a leaky gut are well understood (for details, see my first book, The Paleo Approach where each of these are discussed in detail). But are these what cause the leaky gut in autoimmune disease? Maybe and maybe not.
New insights from this study
Right from the get-go, this study peaked my interest. In this experimental model of multiple sclerosis, healthy mice are inoculated with a chemical called myelin oligodendrocyte glycoprotein. It takes them 2 weeks to fully develop experimental autoimmune encephalomyelitis (EAE) and only about 80% of the mice do develop the disease. The animals that are going to develop EAE characteristically lose weight, about 10% of their body weight, about 1 week after the inoculation (they typically start to show symptoms about 8-10 days after with full development of the disease at the 2-week mark).
What the researchers did was to select those animals who were developing EAE based on whether or not they lost weight at the 1-week mark. Then, they measured intestinal permeability (whether or not the gut is leaky) at 1 week (before development of the disease) and at 2 weeks (after the disease had fully developed). What they found was that a leaky gut was present at 1 week, before the disease fully develops. A leaky gut comes first.
The researchers further characterized exactly what type of damage was being done to the gut to make it leaky as well as exactly how the immune system was being stimulated in the gut. The same types of immune system cells known to be culprits in the damage to the myelin sheath in EAE were accumulating in the tissues of the gut and causing damage there too. And regulatory T-cells, which are supposed to reign in the immune system but are deficient in autoimmune disease, were low in the intestine. Even more interesting, the protein zonulin, which has been implicated as the protein stimulated by gluten that causes a leaky gut via direct action on the tight junctions in celiac disease, was increased (again, lots more details on the effects of zonulin and the roles of different immune cell types in autoimmune disease can be found in The Paleo Approach). Taken together, this all looks very much like a gut being attacked by a dysfunctional immune system.
So what’s going on? This is a model of multiple sclerosis. How is the gut being attacked if this is a disease in which the immune system targets myelin sheaths?
The researchers did something very clever to begin to answer this question (of course, they were clever up to this point too, but this next experiment is what got me all excited). They wanted to test if the immune system was what was damaging the gut and causing a leaky gut in advance of the development of EAE. So, they took immune cells from mice who had developed EAE and put them into healthy mice. Two weeks after the healthy mice had been injected with these immune cells, they had developed leaky guts with all the same markers of inflammation and with increased zonulin.
Aha! The immune system is the culprit! It attacks the tissues of the gut in addition to the targeted attack on other tissues in autoimmune disease!!!
Um, wait a minute.
Then what gets the immune system all fired up in the first place?
I was asked if this paper confirms what I’ve been saying all along: that a leaky gut contributes to autoimmune disease. But, this research shows something altogether more intriguing: that once the immune system develops the ability to attack tissues of the body, the gut is the first victim. The situation is a little bit different than the targeted attack of the immune system on specific tissues (like the myelin sheaths in the case of multiple sclerosis) that happens in autoimmune disease. Instead, the immune system is getting ramped up and, likely because so much of the body’s immune system is housed in the gut, the gut is the first tissue to be harmed by a dysfunctional immune system. Rather than a leaky gut causing the dysfunctional immune system that leads to autoimmune disease, this research shows that it might just be the other way around.
New questions we have to ask
If this research exonerates other factors that can cause a leaky gut in the pathogenesis of autoimmune disease, it then begs the question: if leaky gut isn’t contributing to autoantibody formation and overstimulation of the immune system in autoimmune disease, what is?
Clearly, genetic predisposition is a factor here: it accounts for one third of your risk for autoimmune disease. But, autoimmune diseases are not genetic diseases, and our genes don’t take all of the blame.
In The Paleo Approach, I detail the essential role that many nutrients play in the immune system, nutrients that are sorely lacking in the Standard American Diet. I believe nutrient-deficiency is one of the culprits and there are hundreds of studies pointing to specific nutrient deficiencies in specific autoimmune diseases (many detailed and referenced in The Paleo Approach).
I think the health of our gut microbiome is another clear contender for “thing that can go wrong that can cause immune system dysfunction leading to autoimmune disease”. We know that the beneficial microorganisms that live in our digestive tracts modulate our immune systems. We also know that many foods can support the growth of the wrong kinds of microorganism or the wrong numbers or locations (all forms of gut dysbiosis, also known to exist in every autoimmune disease in which it has been investigated), so we lose that important regulation control on the immune system.
We also know that inadequate sleep (which most of us suffer from), sedentary lifestyles, and chronic stress suppress the immune system. In fact, our modern, Western diets and comfy lifestyles by themselves create the perfect storm of circumstances for immune dysfunction to take place. And this is without the contribution from toxins and pollutants, which are contributing factors too.
It’s also important to note that this study does not preclude a leaky gut caused by some other factor, leading to a dysfunctional immune system, which then attacks the gut first in the development of autoimmune disease. I rather suspect that there are so many contributors to the development of autoimmune disease and that sequencing them definitively may prove impossible.
How does this impact the autoimmune protocol?
This is the big question. The diet and lifestyle recommendations in The Paleo Approach are centered around a few overarching principles: nutrient-density to support immune system regulation, avoidance of immunes-stimulating compounds in foods, avoidance of foods that are gut irritants or damage the gut barrier, avoidance of foods that feed gut dysbiosis, addition of foods that support a healthy gut microbiome, regulation of important hormones known to be immune regulators (like leptin and insulin), and prioritization of lifestyle factors known to influence the immune system (generally through hormonal effects).
Overall, the recommendations target immune regulation. These recommendations also support gut health, i.e., the restoration of the integrity of the gut barrier and the normal diversity, numbers, and location of gut microorganisms. But, the recommendations do not come from the primary goal of fixing a leaky gut. The primary goal is regulating the immune system. And what this study now shows is that by regulating the immune system, that’s one more way this protocol can help fix a leaky gut!
This is good news for those of you using the autoimmune protocol and The Paleo Approach to manage autoimmune disease. This doesn’t change the protocol. It does however put even more emphasis on nutrient-density (I know you don’t like to hear it, but it really really really is important to eat organ meat, seafood, and tons of veggies as the foundation of your diet), not that there was a shortage of emphasis before this. It perhaps puts even more focus on lifestyle factors, most notably getting sufficient sleep (at least 8 hours per night, every single night) and reducing stress. And the good news is, that it means that once we get our immune systems under control, our leaky guts should fix themselves.







 TPV Podcast, Episode 110: Leptin
TPV Podcast, Episode 110: Leptin